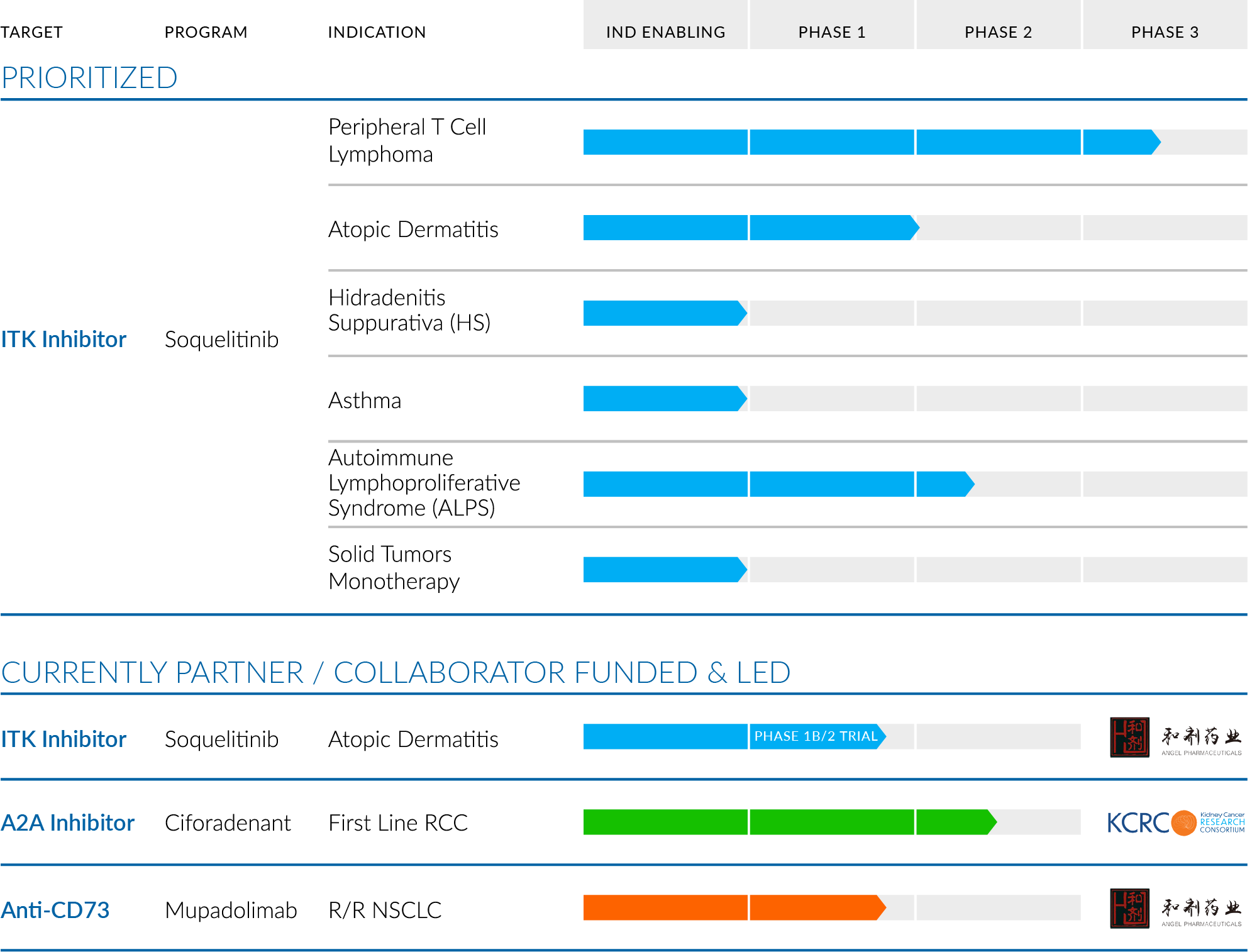
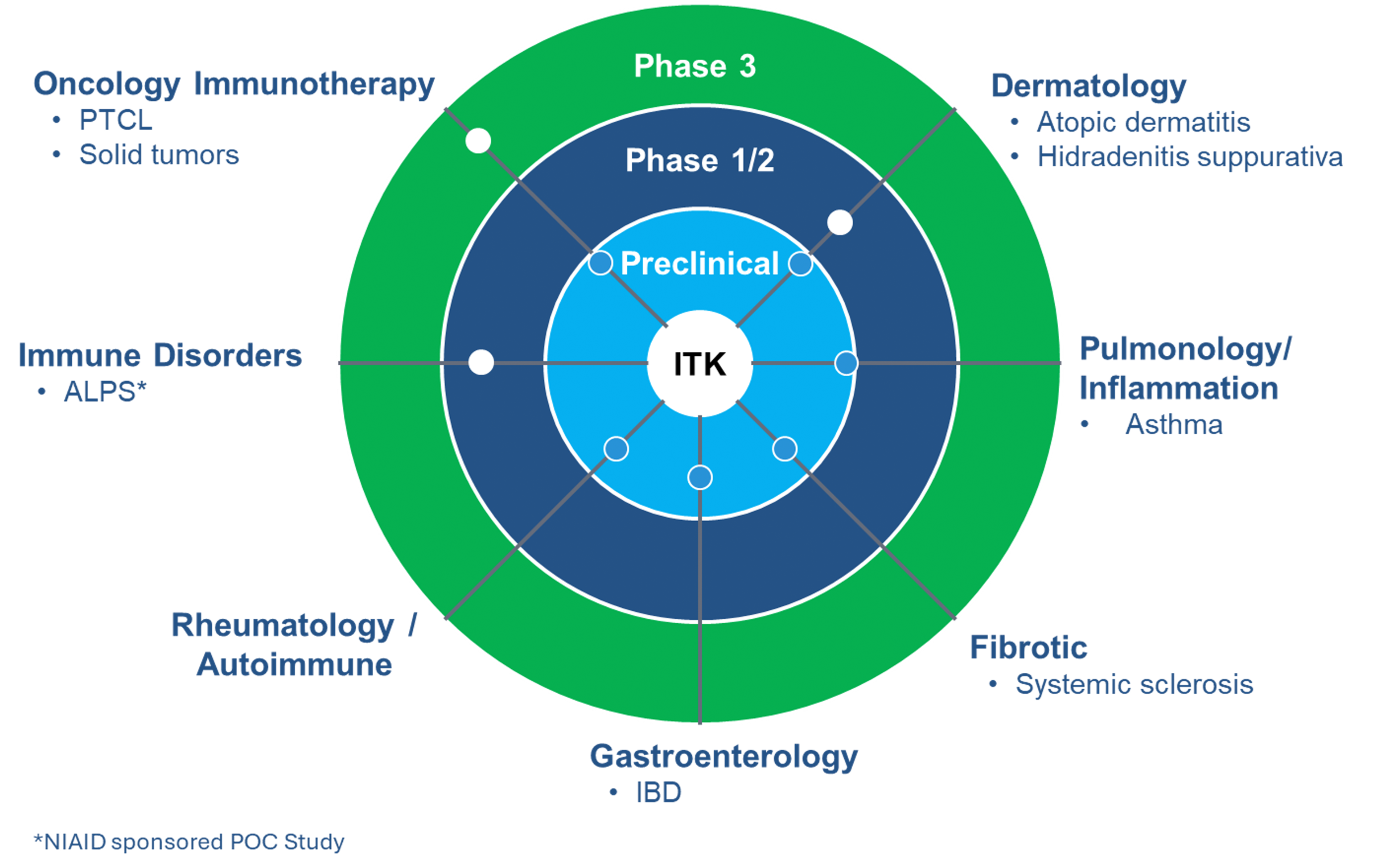
We have multiple clinical-stage programs for immune diseases and cancer with our lead ITK inhibitor, soquelitinib. We also have other programs with differentiated mechanisms of action, providing additional opportunities to deliver new medicines to patients.
Soquelitinib (CPI-818)
Soquelitinib is an investigational small molecule drug given orally designed to selectively inhibit ITK (interleukin-2-inducible T cell kinase), an enzyme that is expressed predominantly in T cells and plays a role in T cell and natural killer (NK) cell immune function. Soquelitinib has been shown to affect T cell differentiation and induce the generation of Th1 helper cells while blocking the development of both Th2 and Th17 cells and production of their secreted cytokines. Th1 T cells are required for immunity to tumors, viral infections and other infectious diseases. Th2 and Th17 helper T cells are involved in the pathogenesis of many autoimmune and allergic diseases. The Company believes the inhibition of specific molecular targets in T cells may be of therapeutic benefit for patients with cancers, including solid tumors, and in patients with autoimmune and allergic diseases. Recent studies have demonstrated that ITK controls a switch between the differentiation of Th17 proinflammatory cells and T regulatory suppressor cells. Inhibition of ITK leads to a shift toward T regulatory cell differentiation, which has the potential to suppress autoimmune and inflammatory reactions.
ITK: Focused on Target and Broad Indication Potential
Ciforadenant (CPI-444)
Ciforadenant is an investigational small molecule, oral, checkpoint inhibitor designed to disable a tumor’s ability to subvert attack by the immune system by blocking the binding of adenosine to immune cells present in the tumor microenvironment. Adenosine, a metabolite of ATP (adenosine tri-phosphate), is produced within the tumor microenvironment where it may bind to the adenosine A2a receptor present on immune cells and block their activity. Ciforadenant has been shown to block the immunosuppressive effects of myeloid cells present in tumors and preclinical studies published in 2018 demonstrated synergy with combinations of anti PD1 and anti-CTLA4 antibodies.
Mupadolimab (CPI-006)
Mupadolimab is a unique anti-CD73 antibody that blocks the production of adenosine in the tumor microenvironment. It also binds to B cells resulting in their activation and differentiation into antibody producing plasma cells. These attributes have the potential to augment both cellular and humoral immunity to cancers. Corvus has completed a phase 1 study in cancer patients and mupadolimab is now being evaluated in non-small lung cancer by its partner in China, Angel Pharmaceuticals.
For more information on our pipeline, please see our Annual Report available in the SEC Filings section of the website.
Our Science
We are developing ITK inhibition as a new approach to control the immune system in order to treat immune diseases and cancer
Join Our Team
Contribute your talent, expertise and desire to learn here at Corvus, where you can help improve the lives of people with immune diseases and cancer